LIHU‘E — A federal team of 30 civilians is being deployed to Hawai‘i to administer monoclonal antibody therapy to treat COVID-19 patients, announced the state Department of Health earlier this month.
Increasing access to monoclonal antibody therapy will decrease severe illness in individuals recently infected with COVID-19, providing relief to Hawai‘i’s hospitals, the DOH said.
Monoclonal antibody therapy, which is not a substitute for vaccination, helps the immune system temporarily respond to the immediate infection.
The U.S. Food and Drug Administration issued an emergency use authorization for a number of monoclonal antibody therapies that serve as early treatment after a COVID-19 diagnosis.
“There is one evidence-based early treatment for COVID-19,” said Dr. Janet Berreman of the DOH Kaua‘i Health District Office during a question-and-answer segment the county’s COVID-19 briefing update Thursday.
“That treatment is monoclonal antibody, or ‘regeneron.’ This is available to high-risk people who have been diagnosed with COVID-19, and in some cases to those who have been exposed to a person confirmed to have COVID-19. It is not available for children younger than 12 years old, or for people who weigh less than 90 pounds. The medication needs to be administered with a physician’s order and under medical supervision. It is either an intravenous infusion or subcutaneous injections. It is available at the Wilcox Medical Center, Ho‘ola Lahui Waimea Clinic and the Samuel Mahelona Memorial Hospital,” she said.
Berreman said this monoclonal antibody therapy program was put in place early during the COVID-19 pandemic.
“Targeted use of monoclonal antibodies could keep Hawai‘i COVID-19 patients from developing severe illness that requires hospitalization,” said DOH Director Dr. Elizabeth “Libby” Char in the DOH announcement.
“However, COVID-19 vaccination remains the most effective way to create long-lasting immunity and prevent severe illness and death,” said Char. “Increasing access to monoclonal antibody therapy will reduce strain on our hospitals. We thank our state and federal partners for their assistance.”
A Wilcox Medical Center spokesperson said the hospital is providing COVID-19 monoclonal antibody therapy to eligible patients in accordance with the FDA emergency use authorization criteria.
“Monoclonal antibody therapy is being used for the treatment of mild-to-moderate COVID-19 in adults and pediatric patients ages 12 and older who meet specific criteria, including health history and duration of COVID-19 symptoms,” the spokesperson said.
“The therapy, which is administered as an outpatient treatment, has been shown to decrease the risk of illness progression and hospitalization.”
Cheryl Tennberg, the Hawai‘i Health Systems Corporation Kaua‘i Region chief nurse executive, said the collaborative effort among the health-care providers offers patients diagnosed with COVID-19 an option toward minimizing hospitalization, and possibly death.
“We are taking yet another strategic step to preserve hospital capacity during a time of unprecedented demand,” said President and CEO of the Healthcare Association of Hawai‘i Hilton Raethel. “Increasing the number of COVID-positive residents who receive this therapy will result in a reduction of symptoms, and reduce demand on our hospitals.”
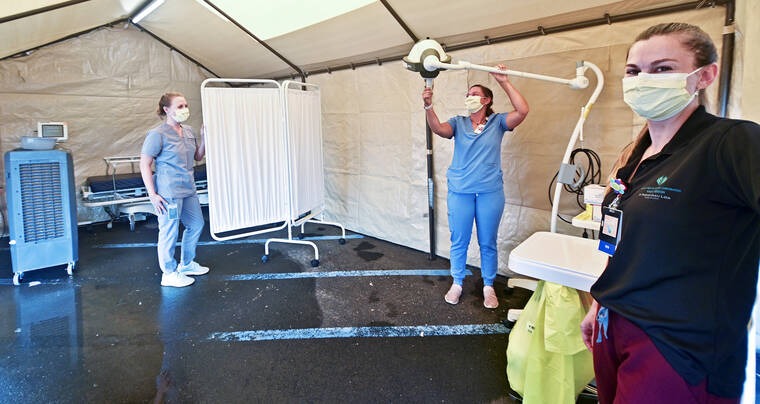
The Federal Emergency Management Agency is providing the 30 clinicians to augment the existing capacity to administer this treatment. They are expected to arrive in Hawai‘i today.
Six teams will be stationed at hospitals or federally qualified health centers across the state, administering treatments seven days a week. The teams will administer monoclonal antibodies by injection. Treatment requires monitoring and the ability to respond to reactions or other medical events.
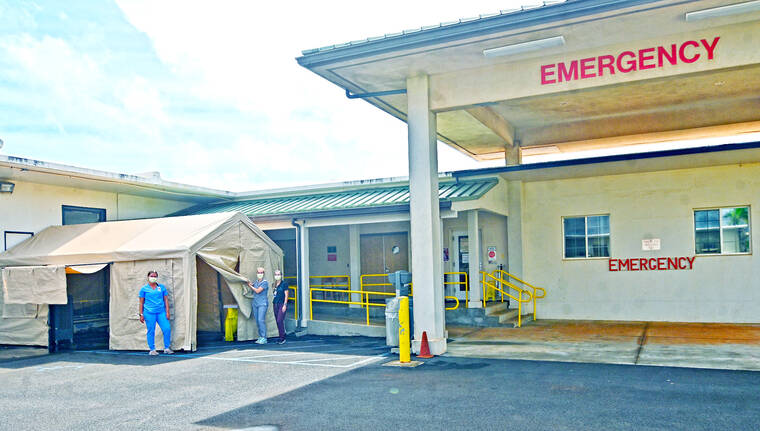
A Wilcox Medical Center spokesperson said there are no FEMA personnel scheduled for Kaua‘i.
However, the Kaua‘i healthcare community is working to provide monoclonal antibody therapies to eligible patients on island seven days a week.
“On the Eastside, the Samuel Mahelona Memorial Hospital offers the therapy,” Tennberg said. “Wilcox Medical Center takes care of the central portion of the island, and Ho‘ola Lahui in Waimea uses facilities on our Kaua‘i Veterans Memorial Hospital campus to offer the service on Monday, Wednesday and Friday to people on the Westside. All of the treatments are by schedule or appointments.”
Wilcox Medical Center officials said they working to provide better access for patients, including a better-operating website for self-scheduling.
Monoclonal antibody therapy is not a substitute for vaccination, the DOH said. Monoclonal antibodies provide a short burst of immunity, compared to the longer-lasting immunity created by vaccination. COVID-19 vaccines are safe and effective at preventing severe illness and death, the DOH said.
Treatment with monoclonal antibodies within the first 10 days of symptom onset could reduce the risk of severe illness, hospitalization and death.
This treatment is authorized for individuals 12 and older with mild-to-moderate COVID-19 who are at high risk for severe illness, not hospitalized and do not require supplemental oxygen. Patients must be referred by a physician. While not everyone will be eligible for monoclonal antibody therapy, all people 12 and older are eligible to be vaccinated.
“To learn more about what these therapies are, and how they can prevent mild-to-moderate symptoms from getting worse, please visit combatCOVID.hhs.gov or talk to your doctor,” a Wilcox Medical Center spokesperson said.
This story was updated on Wednesday, Sept. 22, 2021, for clarity.
•••
Dennis Fujimoto, staff writer can be reached at 245-0453 or dfujimoto@thegardenisland.com.

Thank God, it’s about time, the vaccines sure aren’t working
Vaccines are working fine, its the unvaccinated not working! Don’t know why you vaccinated would accept this science, same as the old science they have blindly rejected like lemmings. Get vaccinated and save us all a heap of trouble and money!
I meant unvaccinated will not accept this science
As the kids say, “Yessah, blessah!”
Thank FLA Gov Ron DeSantis for being early and on top of this for his residents so the rest of the country could watch and learn. Like always, HI ranks last in service to its residents.
MAMA DOES NOT KNOW BEST
Florida surpasses 50K COVID-19 deaths after battling delta wave
By Terry Spencer and Adriana Gomez LiconPublished 4 days ago Coronavirus in Florida Associated Press
MIAMI, Fla. – Health officials say the state of Florida has surpassed 50,000 coronavirus deaths since the pandemic began. The U.S. Centers for Disease Control and Prevention has tallied 50,811 deaths after adding more than 1,500 COVID-19 deaths provided by state authorities on Thursday.
Gov. Ron DeSantis said the latest wave was “really rough,” striking younger and healthier people, including numerous police officers and firefighters.
More than one-fourth of the total COVID-19 deaths in the state occurred this summer as the state battled a fierce surge in infections fueled by the delta variant.